Neuroinflammation and progressive neuronal death are two critical hallmarks of Alzheimer’s disease (AD). Still, the particular mechanisms underlying these alterations have not been fully characterized. To date, researchers have mainly focused on deciphering the role of pro-inflammatory microglia; however, mounting evidence points to a more complex scenario with intricate interconnections between dysfunctional glial cells and dying neurons.
In a new study by the group "Signaling and Cell Damage in Cancer" published in Translational Neurodegeneration with Cristina de Dios as first author and coordinated by Anna Colell, cholesterol is uncovered as a regulatory factor of the immune response in AD mediated by the inflammasome, a molecular platform that integrates different pro-inflammatory signals.
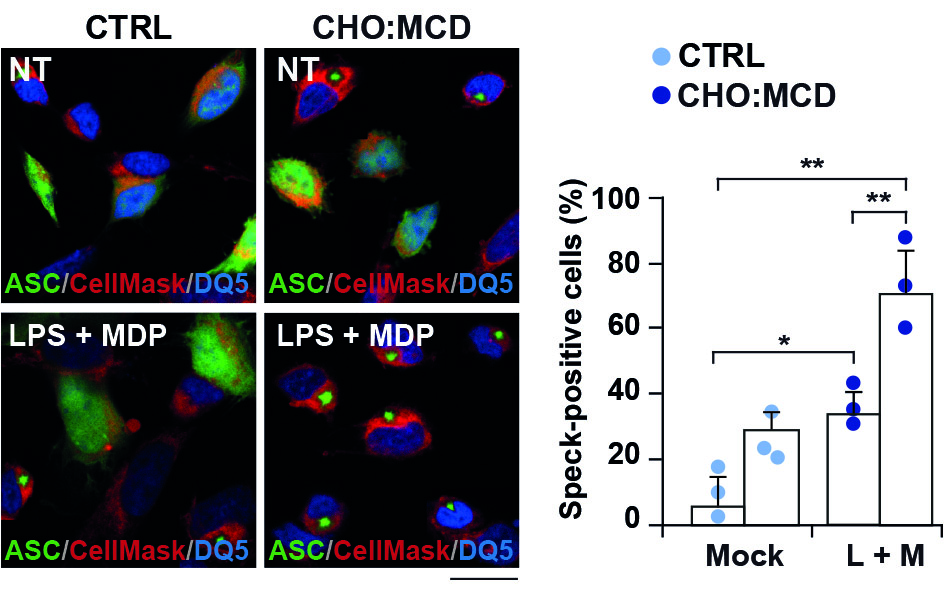
The study shows that an increase in intracellular cholesterol levels stimulates the assembly of the inflammasome, in response to both typical innate immune response inducers (bacterial endotoxins) and amyloid beta peptides. In microglia, this activation is accompanied by the transition to a neuroprotective phenotype, displaying enhanced phagocytic capacity and neurotrophin release. In contrast, induction of the inflammasome in cholesterol-enriched neurons triggers a gasdermin D-mediated pro-inflammatory death known as pyroptosis. Furthermore, incubation of activated microglia with conditioned media from pyroptotic neurons impairs their function, reducing their phagocytic capacity.
This research reveals the key role of cholesterol as a checkpoint of neuroinflammation in AD, regulating the signaling pathways driven by inflammasome differentially in microglia and neurons, thus favoring a neuroprotective phenotype in microglia ultimately challenged by a pro-inflammatory neuronal death.
Paper: de Dios, C., Abadin, X., Roca-Agujetas, V. et al. Inflammasome activation under high cholesterol load triggers a protective microglial phenotype while promoting neuronal pyroptosis. Transl Neurodegener 12, 10 (2023). https://doi.org/10.1186/s40035-023-00343-3