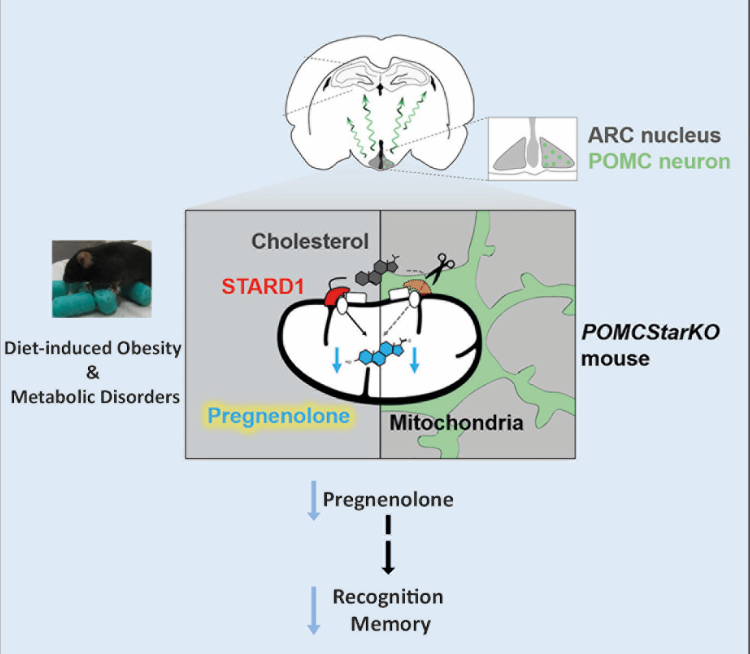
STARD1 is a protein responsible for the transport of cholesterol to the inner mitochondrial membrane for metabolism. In the liver, the mitochondrial metabolism of cholesterol by CYP27A1 represents an alternative route in the synthesis of bile acids that plays a fundamental role in the development of hepatocellular carcinoma, while in steroidogenic tissues, and in hypothalamic neurons (POMC), the transport of mitochondrial cholesterol by STARD1 is a source of pregnenolone, whose contribution to cognitive function has not been established to date. For the elucidation of the functions of STARD1, the generation of genetic models with specific deletion of STARD1 in certain cell types has been essential, since the global elimination of STARD1 is lethal due to adrenal dysfunction and inability to generate steroidogenic hormones. To this end, IIBB researchers from the "Mitochondrial regulation of cell death" group led by Jose C Fernandez-Checa and with the participation of Carmen Garcia Ruiz and Vicent Ribas have recently generated a genetically modified mouse in which the STARD1 gene has been marked with Lox P sites (Stard1f/f mice) for its specific ablation in certain cell types.
This model of Stard1f/f has been fundamental for its specific deletion in POMC neurons of the hypothalamus (POMCStard1KO) developed by Marc Claret, leader of the "Neuronal control of metabolism" group at IDIBAPS in collaboration with the IIBB researchers, published in Cell Metabolism. The metabolic studies carried out by Marc Claret's group showed that STARD1 determines the physiological level of pregnenolone in POMC neurons, and that both the treatment of high-fat diets (DRG), which cause a metabolic and functional alteration in the hypothalamus, like the POMCStard1KO model exhibit a decrease in the concentration of pregnenolone, a neurosteroid essential for cognitive function. Cognitive dysfunction in DRG-treated as well as POMCStard1KO mice is reversed after intracerebroventricular administration of pregnenolone. The relevance of these studies is highlighted in a study of a cohort of obese subjects. The determination of pregnenolone in the cerebrospinal fluid of obese patients showed a correlation with cognitive impairment (Mini-Mental Status Examination, MMSE). These studies therefore demonstrate for the first time the connection between cholesterol metabolism in the hypothalamus in the context of metabolic disorders, and open the therapeutic pathway of neurosteroids, in particular pregnenolone, for the treatment of cognitive dysfunction associated with obesity. This research has been multidisciplinary with the participation of various CIBER groups, including CIBEREHD, CIBERNED, CIBERER, CIBEROBN and CIBERDEM.
Referencia del artículo:
Ramírez S, Haddad-Tóvolli R, Radosevic M, Toledo M, Pané A, Alcolea D, Ribas V, Milà-Guasch M, Pozo M, Obri A, Eyre E, Gómez-Valadés AG, Chivite I, Van Eeckhout T, Zalachoras I, Altirriba J, Bauder C, Imbernón M, Garrabou G, Garcia-Ruiz C, Nogueiras R, Soto D, Gasull X, Sandi C, Brüning JC, Fortea J, Jiménez A, Fernández-Checa JC, Claret M.
Hypothalamic pregnenolone mediates recognition memory in the context of metabolic disorders. Cell Metab 2022 Feb 1;34(2):269-284.e9.
doi: 10.1016/j.cmet.2021.12.023